The potential benefits of SFC gained momentum through the 1990s where both academia and industry regarded the technique as an invaluable separation tool both in analytical and preparative separation. However; this was short-lived since challenges with reproducibility and hardware stability reduced confidence in using SFC as a mainstream analytical tool. It continued to maintain a force within the preparative arena since both chiral and achiral compounds could be isolated in the absence of an aqueous mobile phase. In addition, since carbon dioxide is gaseous at atmospheric pressure, the primary mobile phase constituent is simply separated from the compound of interest via a gas-liquid phase separator (GLS).
The recent development of modern instrumentation to overcome the challenges faced by earlier systems has started to rebuild the confidence with this technique. This has led to a renaissance with an explosion in academic papers and adoption within both the pharmaceutical and biopharmaceutical arena. Furthermore, new application areas have begun to trend; such as food science, natural products, and environmental analyses are all utilizing the benefits of modern SFC analysis.
The reduced extra-column volumes within instrumentation coupled with the successful packing of sub-2µm superficially porous particle stationary phases have led to ultra-high performance SFC (UHPSFC) facilitating improved resolution and faster analyses. The ability to hyphenate to mass spectrometry via a split-less interface has further propelled this technique into new application paradigms beyond the pharmaceutical world; one of which includes the Food and Environmental Testing Industry.
Application example
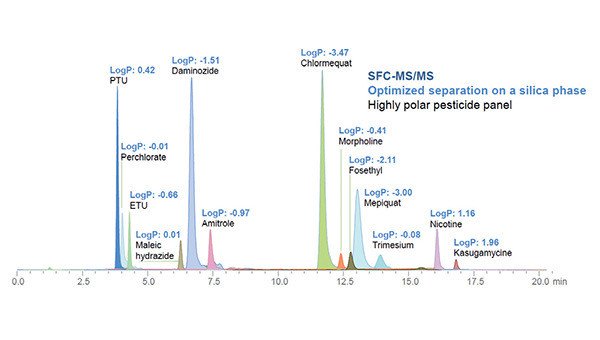
New advances in LC-MS/MS capabilities have led to the development of sophisticated multi-residue methods measuring hundreds of compounds in a single method. Typically, methods utilize reversed-phase chromatography which can result in a small but significant number of compounds not retained by C18 stationary phase columns due to the highly polar nature of some compounds. This is a common challenge observed by many industry sectors, one of those being food safety. The quantification of highly polar pesticides in a range of food matrices is notoriously challenging. The tolerated solution involves the use of more exotic column stationary phases which can suffer batch-to-batch challenges and %RSD variation over a relatively small number of injections due to the build-up of matrices within the stationary phase bed.
An alternative approach utilized supercritical fluid chromatography and a mixed-mode ternary gradient which provided superior retention of highly polar pesticides over reversed-phase strategies.
Properties of supercritical fluids
Supercritical fluids show certain properties that are used to advantage in chromatographic separations:
- No liquid/gas phase boundary and therefore no surface tension
- Solute solubility increases with an increase in fluid density
- Fluid density increases with pressure, therefore solute solubility increases with the pressure
- At constant density, solute solubility increases with temperature
- Solvating power like organic solvents but higher than those of gases
The solvating power of a supercritical fluid is proportional to its density, which in turn can be increased by increasing the system pressure.
Advantages of SFC
Supercritical fluids show low viscosities and higher diffusivities when used as a mobile phase. This results in narrower peaks because of the rapid diffusion and faster elution with a lower pressure drop through the column. SFC can be used with a wide range of detectors and has a resolving power that is 5 times that of HPLC.
Author: Dr. Raymond Wong is LC Product Manager at Shimadzu UK